You may have read and/or seen my recent assignment: 8-4-08: Eight stories that I think will break on the beat in '08. Here's an inside scoop: CNBC editorial managers like the franchise because it's forward-looking, which is, of course, what investors want.
But this time of year you see a lot of retrospective pieces in the mainstream media. (Last night while channel surfing -- okay, occasionally I'm ashamed to admit I get sucked into watching them -- one of the entertainment shows was doing a piece on 2007 Hollywood couple breakups.)
So, not to be left out, here's an off-the-cuff take -- in no particular order -- on what I think were the seven biggest stories in my sectors in '07.
1. Avandia. The Dr. Steven Nissen study published in "The New England Journal of Medicine" last May ignited a firestorm over the safety of the GlaxoSmithKline diabetes drug, and a medical and scientific community debate about the validity of meta-analysis. That's when researchers collect data from a bunch of clinical trials or medical records and try to draw conclusions.
2. Dendreon. The saga of the prostate cancer drug, Provenge, is one of the richest stories I've covered in my five years or so following biotech and pharma. The dramatic and emotional advisory committee meeting, the subsequent FDA delay of the drug's approval and now a request for a congressional investigation. Making no judgements whatsoever on the drug or the stock, this is simply a great story from a reporter's point of view.
3. Pfizer pulling the plug on the inhaled diabetic insulin Exubera.
For the world's biggest drug company to invest all those years and all that money on what was originally hailed as a revolutionary breakthrough -- and then, only months after getting it to market, deciding to throw in the towel because it wasn't selling? Remarkable, to say the least.
Some analysts applauded Chief Executive Jeff Kindler for grabbing the bull by the horns and making such a bold call. Others saw it as emblematic of the problems at Pfizer.
4. The failure of Biogen Idec to find a buyer.
The biotech company put itself up for sale, the stock shot to the moon and then no one wanted to pay that much money for the company. Did big pharma draw a line in the sand? Did Carl Icahn lose his touch? Good questions for '08.
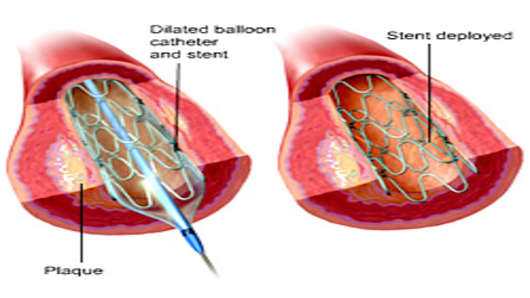
5. The stent controversy. This one plays into the populist opinion that one day one study says one thing, the next day another study says the exact opposite, etc.
First, we saw studies saying drug-coated stents may cause fatal blood clots. Then a landmark study came out last March suggesting plain-old heart drugs may work better than stents -- bare metal or drug-coated. Then there were more conflicting studies about drug-coated versus bare metal stents. And against that backdrop we saw two companies -- Abbott Laboratories and Medtronic -- go before FDA Advisory Committees to try to get new drug-coated stents on the market.
6. Amgen. The world's biggest biotech company by sales announces major cutbacks to deal with plummeting revenue for its anemia drug franchise.
Aranesp and Epogen come under intense pressure due to safety concerns and Medicare and insurance reimbursement issues. The worry is that patients who are given too much of the drugs could die or see their tumors grow. The FDA issued severe safety warnings and new treatment guidelines were developed. This year the stock has gone from nearly 80 dollars to 47 and change. Wow.