Shares of Biogen and Eli Lilly fell Wednesday after highly anticipated updates on the company's Alzheimer's programs.
Both drugs aim to clear amyloid plaque buildups from the brain in an effort to slow Alzheimer's cognitive declines. The drugmakers said Wednesday both showed signs they may do that. Lilly's drug displayed consistently better results the longer patients took its drug, while Biogen's showed it yielded stronger efficacy the higher the dose.
While Biogen called its update encouraging, investors were hoping a closely watched middle dose would significantly clear amyloid plaques from the brain and slow Alzheimer's cognitive declines. In the update Wednesday, it individually succeeded on the first, but didn't show statistical significance on two measures of cognition. The stock was down 4.8 percent in early morning trading. (For a real-time stock quote, click here.)
Lilly shares fell 3.3 percent in early trading. (For a real-time stock quote, click here.)
Overall, Biogen Chief Medical Officer Alfred Sandrock said, the 166-patient trial showed the medicine showed efficacy on both measures over the course of the year, and that it increased the higher the dose.
Read MoreCNBC Explains: New Alzheimer's drugs
"The data are completely consistent with and only reinforce what we saw back in March," Biogen Chief Medical Officer Alfred Sandrock said in a telephone conversation, referring to results that drove the company's stock up 11 percent as investors hoped the company was onto a drug that could modify the course of the disease.
But some were skeptical. Piper Jaffray downgraded Biogen's stock on the data, calling the results sloppy in a note Wednesday.
"We do not believe this is an attractive pipeline program," Piper analyst Josh Schimmer wrote. He lowered his price target on the stock to $410 from $485.
Other analysts were more supportive, with Cowen and Evercore ISI maintaining their predictions of 50 percent probability of success for the program. That's a much higher chance than history has suggested: since 1998, more than 120 programs in Alzheimer's have failed, according to industry group PhRMA. Just four drugs were approved, none targeting the disease's underlying biology.
Read MoreWomen hurt by Alzheimer's twice as fast
Alzheimer's disease afflicts more than 5 million Americans, a number expected to triple by 2050 without effective interventions. Analysts say a successful medicine for the memory-robbing disease could draw $20 billion annually.
So promising drugs in development from both Biogen and Eli Lilly are capturing Wall Street's attention. Updates came Wednesday during the Alzheimer's Association International Conference in Washington, D.C..
Analysts had hoped the dose of Biogen's drug, aducanumab, of 6 milligrams per kilogram of patients' weight, would show stronger efficacy than two lower doses reported in March, and fewer side effects than the highest. Sandrock said the analysis of all doses in the trial showed higher ones did yield greater effects on both plaque clearance and cognition even if each individual, smaller arm did not.
"With these clinical endpoints you're talking about 25-30 patients per group," Sandrock said. "It stands to reason it's going to be a little jittery about where they land."
The company has begun screening patients for two large late-stage studies beginning in 2015.
Biogen reported data in March on patients who had taken the other doses in the trial, of 1, 3 and 10 milligrams per kilogram for about a year. Patients in the 6-milligram group had only been on the drug for about six months at that point, making Wednesday's data release a focal point for investors.
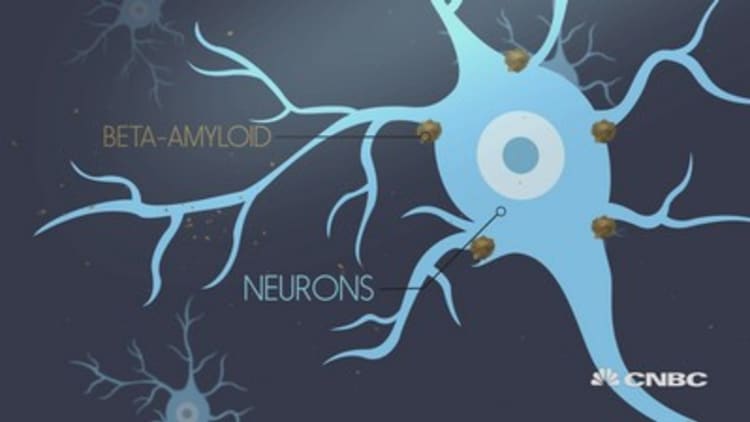
In the trial, Biogen measured cognition on two scales: MMSE, or Mini Mental State Examination, and CDR-SB, Clinical Dementia Rating sum of boxes. They're tests designed to assess everyday mental skills and recall, with points assigned to answers.
On MMSE, patients taking placebo declined by 2.81 points after a year. That compared with a decline of 2.18 points for the 1 milligram dose, 0.7 points for the 3 milligram, 1.96 points for the 6 milligram and 0.56 points for the 10 milligram. Biogen said only the results for the 3 and 10 milligram doses individually reached statistical significance.
On the CDR-SB measurement, patients on placebo declined by 1.87 points on average, versus 1.72, 1.37, 1.11 and 0.63 for the 1, 3, 6 and 10 milligram doses, respectively. On this scale, only the 10 milligram dose individually showed statistical significance.
Safety was also closely watched. The trial has shown a side effect known as ARIA, for amyloid-related imaging abnormalities, associated with removal of plaques from the brain. Biogen said in general, ARIA was seen early in the course of treatment, showed either no symptoms or only mild ones, and most patients seeing the side effect continued treatment at a lower dose.
"I don't think this really changed very much," Sandrock said of Wednesday's results. "We're enthusiastic about our phase 3 and we're doing our very best to start enrollment in that trial as soon as possible."
Lilly's Alzheimer's drug
Lilly reported data Wednesday from a follow-on study of two of its earlier trials. The drug, solanezumab, had missed its overall goals in those studies, but showed signs of efficacy among patients in the earlier stages of Alzheimer's. Lilly continued testing the medicine on those patients, and in this analysis, switched the patients who had taken placebo onto the drug as well.
Known as a delayed-start analysis, the study compared patients who started 1.5 years later with those taking the drug all along. In the results reported Wednesday, Lilly showed the patients who started later didn't catch up with those taking the drug for longer, but maintained a consistent difference on measures of cognition.
BMO analyst Alex Arfaei, in a note Tuesday, said investors may hope to see the benefit increase over time, and said the stock may decline slightly on the update.
Lilly said the results support the design of its large late-stage study, expected to conclude next year, which may be the basis of filing for regulatory approval.
(UPDATE: This story was updated to include analyst comments and stock performance after the market opened.)

