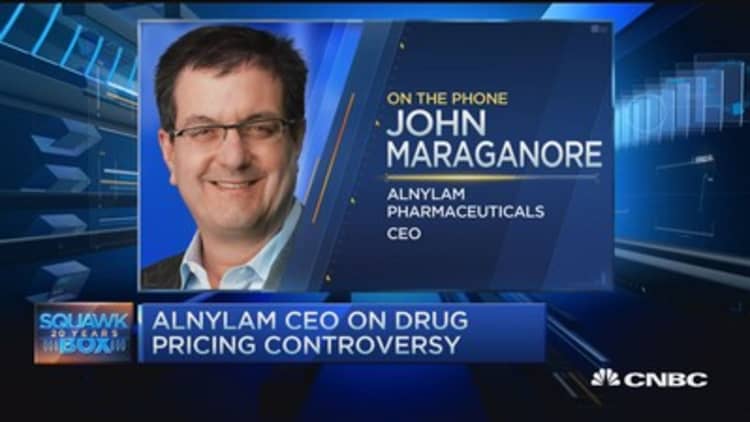

John Maraganore, CEO of biotech Alnylam Pharmaceuticals, said Wednesday his industry focuses on innovation, not pushing prices on decades-old drugs higher to make a profit.
Maraganore was responding to outrage over how Turing Pharmaceuticals, a start-up run by a former hedge fund manager, had jacked up the price of Daraprim, approved by the FDA in 1953, by more than 5,000 percent overnight.
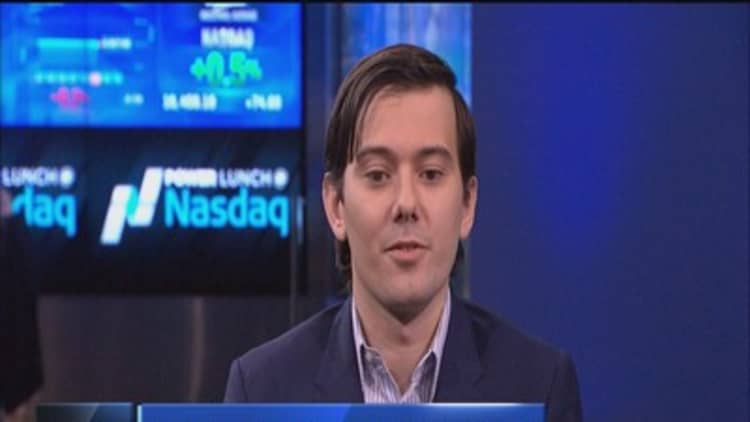
In an interview Monday on CNBC, Turing CEO Martin Shkreli said the drug had been priced too low and his company needed to generate profits to spend on new research and development.
A day later, Shkreli said he would lower the cost of the drug used to fight toxoplasmosis, a parasite infection that can cause serious problems for people with weakened immune systems as a result of AIDS, chemotherapy, or pregnancy. He told NBC News the price, which had been raised to $750 a pill, would return to $13.50 in a few weeks.
Read More Drug-gouging debate isn't CEO's first controversy
"This is not what we do in the biotech industry," Alnylam's Maraganore told CNBC's "Squawk Box" on Wednesday. "We're about innovation, patience and 21st century medicines. We're not about repricing drugs from the 1950s to make a profit. It's not how we focus our R&D investments."
"Most Americans get it when people work hard, they take risks and then they get rewarded. But people do tend to hate it and dislike it when people try to cheat the system," he said.
The outrage sparked by Turing, which bought Daraprim from Impax Laboratories in August for $55 million, also drew the attention of Democratic presidential candidate Hillary Clinton, whose tweet on the issue Monday knocked biotech stocks sharply lower.
Read MoreAllergan CEO fires back on Hillary Clinton tweet
Clinton on Tuesday outlined a plan that she said would limit how much patients have to pay out of pocket for medications each month.
Maraganore said prices for new drugs are not too high. "The focus should be on value and access. There's a lot of data that shows that drugs actually reduce overall costs," he contended—claiming money spent on medications can lower health-care expenditures.
—NBC News and Reuters contributed to this report.


