
The Food and Drug Administration on Friday announced a plan for tobacco and nicotine regulation, which seeks to lower nicotine in cigarettes to non-addictive levels.
This would be just the first step in a sweeping review of the tobacco industry's regulations by the agency, with the goal of encouraging the development of new products that are less dangerous than cigarettes. The FDA has also said it will consider the role of menthol and other flavors in tobacco products.
The statement by the FDA largely came as a surprise and will likely face much resistance from tobacco companies, which are already facing a declining consumer base for their products.
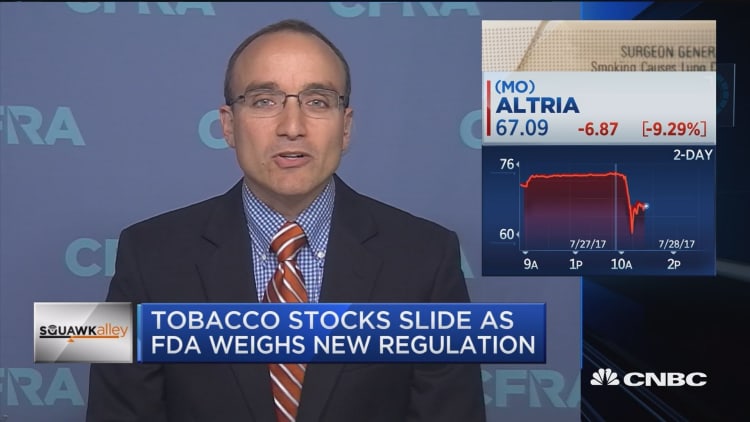
Shares of Altria Group, maker of Marlboro and Parliament brands through its Philip Morris USA unit, plunged more than 10 percent after the FDA's announcement. British American Tobacco, which owns brands such as Camel, Lucky Strike and Newport, fell nearly 9 percent. Vector Group, which sells Liggett Select, Eve and other cigarette brands, saw its stock decline more than 4 percent.
Shares of Philip Morris International, which sells Marlboro and other brands outside the U.S., also dropped nearly 3 percent on the news. Imperial Brands, formerly known as Imperial Tobacco Group, fell 6 percent.
FDA Commissioner Scott Gottlieb called the amount of death and disease attributable to tobacco "overwhelming." Cigarettes are "the only legal consumer product that, when used as intended, will kill half of all long-term users," he said in a statement.
"Envisioning a world where cigarettes would no longer create or sustain addiction, and where adults who still need or want nicotine could get it from alternative and less harmful sources, needs to be the cornerstone of our efforts — and we believe it's vital that we pursue this common ground."
The FDA said Friday that it plans to begin a "public dialogue" surrounding lowering nicotine levels by way of "achievable product standards." The agency added that it's prepared to issue guidance that will describe a new enforcement policy "shortly."
Altria said, in a statement, that it plans to be "fully engaged throughout the process.
"Dr. Gottlieb's comments regarding nicotine and menthol do not come as a surprise to us," the spokesperson said. "We are well prepared and look forward to participating in a thorough process to develop a comprehensive plan for tobacco and nicotine regulation."
"We believe future success will require transformative, innovative products and changing the conversation about tobacco harm reduction," the company added.
Gottlieb said the agency is planning to look at regulation to "render cigarettes minimally addictive."
The FDA said it also will consider an exemption for what it calls premium cigars, and will consider a delay in implementing new rules for reduced-risk offerings like e-cigarettes.
"We've longed believed the FDA would ultimately take a more comprehensive approach toward regulating nicotine as a natural next step," Wells Fargo analyst Bonnie Herzog said in a note to clients.
"Overall, while the market is viewing today's announcement as a 'negative' for cigarette manufacturers, we believe this could prove to be an opportunity over the long term for reduced risk products and, therefore, a positive for Altria/PM as they have a unique competitive advantage."
One concern remains that though the FDA seeks a balanced approach to avoid "unintended consequences," such as activity in the black market, this might not pan out as anticipated, Herzog said. It also increases the risk that a federally mandated ban on menthol cigarettes would be implemented, she added.
"Surely" this encourages a black market, Joseph Agnese, an analyst at research firm CFRA Research in New York, told CNBC's "Squawk Alley" on Friday. But this will also take awhile to go into any sort of effect, he said.
For now, "these actions are actually supportive of the future demand of e-cigarette products," Agnese said. And regulations would primarily impact younger smokers.
Notably, e-cigarettes and vaping devices that were on the market in August 2016 won't be subject to review until 2021 or 2022, depending on the type of product, the FDA said.
The Campaign for Tobacco-Free Kids, an anti-smoking group, said the plan has "great potential to save lives." However, they were critical of the delayed oversight of these newer tobacco products.
"There is no reason to allow these products to stay on the market while developing and implementing the comprehensive strategy Dr. Gottlieb outlined today," the group's president, Matthew Myers, said in a statement.
WATCH: FDA to seek cutting nicotine levels in cigarettes, causing tobacco stocks to tumble



