It is most definitely brain surgery.
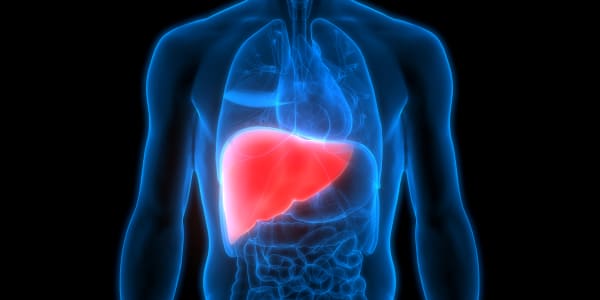
The skull is opened, and electrical leads are implanted directly into the tissue of the brain. These wires are connected to a small device that sends a small dose of electricity into the brain.
The treatment, called deep brain stimulation, means patients suffering from neurological disorders like Parkinson's Disease can have the chance to live a normal life without constantly fighting their own bodies.
Tremors, or involuntary shaking while a limb is at rest, can be noticeably reduced or even eliminated. Patients regain mobility and are able to walk and even talk normally. YouTube is filled with hundreds of videos demonstrating the "life-changing" effects of a DBS device when active, as well as what patient functionality returns to when the device is turned off.
Popularly known as a "brain pacemaker," deep brain stimulation works by blocking errant signals from damaged brain cells to "reset" the brain's natural rhythm, said Dr. Michael Okun, the medical director of the Parkinson's Foundation and chairman for the Department of Neurology at the University of Florida.
During surgery a hole is inserted into the patient's skull and electrical leads are implanted directly into the brain's tissue. The leads are threaded out, and the skull is closed. These wires are threaded behind the ears, down the neck and to the front chest area, where they are connected to a watch-size device that emits a controlled amount of electricity. The entire unit, from wires to generator, are all buried just below the skin.
A growing medical technology niche
The generator, a self-contained unit with a battery, sends pulses of electricity through the leads to stimulate areas of the brain, reducing or in some cases even eliminating the most debilitating parts of the disease. A small, cellphone-size remote control can be used to adjust the "dosage" of electricity and can turn the device on and off.
One of the major manufacturers of the device is Medtronic, which has been active in the U.S. market the longest — it first received approval for use of its DBS system for advanced Parkinson's in 2002; the FDA approved it in 2016 for use with patients who have had Parkinson's for at least four years. Abbott Laboratories acquired the DBS business with its acquisition of St. Jude Medical last year; and Boston Scientific, which currently offers its DBS system in Europe, hopes to receive U.S. approval by year-end.
The three companies' business lines focused on neurology had roughly $1 billion in combined sales in the most recent reported quarter. Medtronic's brain therapies group is the leader, with $522 million in sales in the most recent quarter, though it does not break out DBS sales specifically. Abbott's neuromodulation unit had $300 million in global sales, with approximately 10 percent coming from DBS sales, but it says DBS has been growing since launching in the United States last year.
We're using a tiny amount of electricity to influence a large circuit that is malfunctioning, and we're trying to reset the brain.Dr. Michael Okunmedical director of the Parkinson's Foundation and chairman for the Department of Neurology at the University of Florida
Each DBS surgery can cost between $35,000 and $50,000, and upward of $70,000 to $100,000 for bilateral procedures (both sides of the brain), according to the National Parkinson Foundation. These estimates include the cost of the surgery, devices, anesthesia, hospital fees and physician fees. Device costs can run to tens of thousands of dollars: The Medtronic DBS unit costs $23,000. Since DBS is approved by the Food and Drug Administration for the treatment of Parkinson's Disease, Medicare and most private insurance carriers will cover most, if not all, of the costs of the operation, according to NPF.
Still, as with any surgery, there are risks. "The main risk is bleeding," said Dr. Matthew Wodziak, a neurologist who specializes in Parkinson's Disease at Loyola Medicine. "When we find the right place to put something inside the brain, you have the risk of bleeding [and] infection," he said.
Properly implanted leads do not cause brain damage, and only create microlesions, or small cuts in the brain tissue, and a buildup of glial tissue, cells that support neurons, Okun said.
In addition to the complications associated with surgery, DBS devices also require physical maintenance. Batteries in the devices are not rechargeable and need to be replaced, Wodziak said. The hardware can wear over time and may need maintenance. Medtronic says its devices are not susceptible to hacking, and include encrypted signal technology and proprietary algorithms to communicate with remote controllers.
Despite progress, experts and doctors are trying to determine exactly how electrical stimulation alleviates the symptoms of brain-related disabilities, Okun said. Doctors are still debating if DBS directly affects the function of brain cells, if it simply introduces changes in the chemical composition and biology of the brain, or some combination of the two.
What they do know, Okun said, is that introducing electricity blocks the signals of cells close to the lead while stimulating the "pipes" connecting neuron cells, called axons. It also alters the neurochemistry of the brain, the "ocean" of fluid in which neurons sit.
"We're using a tiny amount of electricity to influence a large circuit that is malfunctioning, and we're trying to reset the brain," Okun said. The electricity restores normal oscillations of the brain, improving the patient's symptoms, he said. In the case of tremors associated with Parkinson's disease, it can make it much more manageable or even stop the tremors completely.
Advances in DBS technology
The treatment can be used to tackle additional neurological problems and other disorders, Wodziak said, including epilepsy, seizures and dystonia, which is the sustained, involuntary contraction of muscles and can lead to painful cramping of the feet or hands, curling toes, or turning and twisting of the neck. A version of the same technology can even be used on the spinal cord. This year, Dr. Andre Machado of the Cleveland Clinic performed the first DBS surgery for stroke recovery using the Boston Scientific Vercise system. It is also considered for depression and weight loss.
Although the treatment has been around since the 90's, there have been some incremental advances to the technology, including leads capable of focusing electricity to increasingly specific parts of the brain and smaller generators capable of connecting to devices using Bluetooth. Abbott's DBS system was the first to be approved in the United States with a "directional lead," which allows physicians to direct current more precisely than traditional DBS. It also allows for system control from an iPod Touch.
Future deep brain stimulators may even include sensors that allow it to respond dynamically to changes in brain patterns, said Brett Wall, senior vice president of brain therapies at Medtronic. The company also hopes to reduce the size of devices, eventually making generators rechargeable or even small enough to embed directly on top of the skull, Wall said.
Researchers also are exploring ways to create a non-invasive form of DBS. MIT scientists have developed a new method to stimulate cells inside the brain non-invasively, using multiple electric fields applied from outside the organ, according to a study conducted on mice that had its results published in the scientific journal Cell in June.
Wodziak cautioned though that the surgery is only optimal for patients that will drastically benefit from it. Due to the nature of the surgery, patients opting for DBS it must undergo a bevy of tests from an army of specialists: A neurologist and neuropsychologist to screen patients. A neurosurgeon to perform the implant. A psychiatrist and rehab team including physical therapists, occupational therapists, and speech therapists to assist recovery.
The best candidates for DBS
Have had PD symptoms for at least five years.
Have "on/off" fluctuations (when a person cycles between "on" time, when the medication is working to control symptoms, and "off" time, when medication has worn off and PD symptoms return), with or without dyskinesia. Dyskinesia is involuntary, irregular, writhing movement that can range from mild to violent and can sometimes be unpredictable.
Continue to have a good response to PD medications, especially carbidopa/levodopa (though the duration of response may be insufficient).
Have tried different combinations of carbidopa/levodopa and dopamineagonists under the supervision of a movement disorder neurologist or specialist.
Have tried other PD medications – such as entacapone, tolcapone, selegiline,apomorphine or amantadine – without beneficial results.
Have PD symptoms that interfere with daily activities.
(Source: National Parkinson Foundation)
Parkinson's disease patients are also advised to consult with a movement disorders specialist, a neurologist who has completed specialized training in PD and movement disorders, or in the least with neurologists in practice who have become experts in this area through their experience caring for many patients and by taking continuing medical education courses.
"We don't put it in first," Okun said. "We very carefully go through an interdisciplinary evaluation, we find out what's disabling to the patient, we have to make a conscious decision; can our technology tackle all the symptoms in our patient?"
The treatment is meant to be a supplement to, and not a replacement for, medication and other treatments. Patients also need to be aware that many post-operative visits for both DBS programming and medication adjustments will be required, most frequently during the first 6 months following implantation. Many medical centers that offer DBS also require monthly visits for the first six months. DBS is removable and reversible.
It's not a cure for Parkinson's Disease.
Michael J. Fox, the actor who has become synonymous with the fight to find a cure for Parkinson's and whose foundation offers guidance for those considering the procedure, has spoken in the past about the potential benefits of DBS, but also its limitations. He was first diagnosed in 1991 and in the late 1990s, Fox had a type of brain surgery to relieve his tremors, a thalamotomy — which destroys a small portion of brain tissue — before DBS became an option.
Fox told CNN's Chief Medical Correspondent Dr. Sanjay Gupta back in 2010 about his decision to not pursue DBS: "The next time they go in my brain, I want them to get it done. … If there is something that they can do that will be curative or restorative … in a much more fundamental way than a mechanical stop gap, a kind of pacemaker. ... That's just my personal preference, but it's been tremendous in people's lives, hundreds I've met, and thousands that have had the procedure, deep brain stimulation. It's been huge, the improvement ... the people that couldn't walk, people that had a hard time functioning."
— By Mike Juang, special to CNBC.com