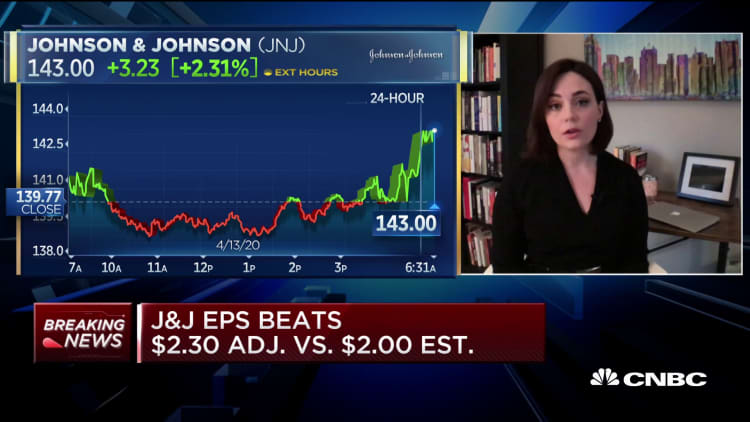
Johnson & Johnson shares rose by more than 3% in premarket trading Tuesday after the drugmaker raised its quarterly dividend — even as it cut full-year earnings guidance due to the coronavirus outbreak.
J&J raised its dividend from 95 cents a share to $1.01, a 6.3% increase, and beat Wall Street's earnings and revenue expectations. On an adjusted basis, J&J reported $2.30 per share during the first three months of the year, higher than the $2 a share expected by Refinitiv analysts. It generated $20.7 billion in revenue, higher than the $19.47 billion expected.
"The dividend this morning is a real good sign of the strength of the company. We're in great financial position," Chief Financial Officer Joseph Wolk said in an interview on CNBC's "Squawk Box" shortly after the earnings were released. "We think this could lend itself to some opportunity for us."
The company lowered its 2020 adjusted earnings forecast to a range of $7.50 to $7.90 per share, from its prior estimate of $8.95 to $9.10 as the Covid-19 outbreak hit the company's performance.
Sales in J&J's medical-device unit fell by 8.2% to $5.9 billion during the quarter as the pandemic forced hospitals to postpone elective surgeries and Americans stayed home.
Wolk noted that the company had forecast a 65% to 80% decline in elective procedures during the second quarter.
"What we saw in the first week was closer to that 65% range," he said.
Wolk said he believes the market is "confident" in the company, adding "we will get through the pandemic as a society." Shares of J&J are up roughly 2% over the last 12 months but down about 4% year to date.
J&J's pharmaceutical business generated revenue of $11.13 billion, an 8.7% year-over-year increase. The company's consumer unit, which makes beauty products such as Neutrogena, generated $3.6 billion in revenue, up 9.2% from a year earlier.
J&J is the first U.S. drugmaker to report earnings post-Covid-19 outbreak, which has infected 1.9 million people worldwide and killed at least 120,449 as of Tuesday morning, according to data compiled by Johns Hopkins University.
Late last month, the company said human testing of its experimental vaccine for the coronavirus will begin by September and could be available for emergency use authorization in early 2021.
J&J also committed more than $1 billion of investment in partnership with the federal Biomedical Advanced Research and Development Authority, which is part of the Department of Health and Human Services, to co-fund vaccine research.
During a Tuesday post-earnings conference call, J&J executives said they expect to produce between 600 million and 900 million doses of the potential vaccine by the end of the first quarter of 2021 if human testing goes as planned.


