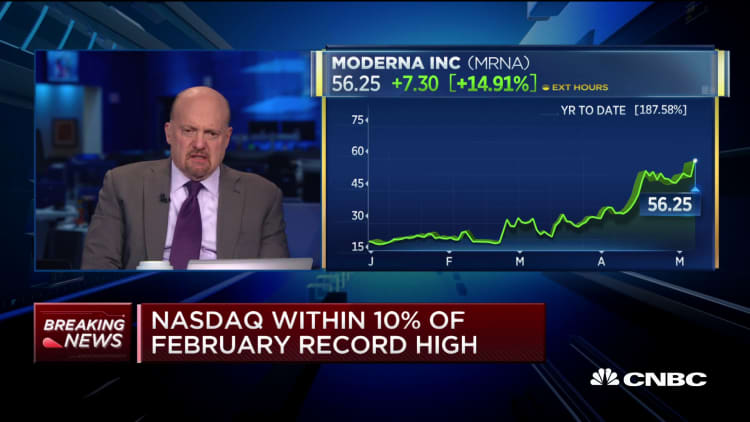
CNBC's Jim Cramer said Thursday he's gained confidence in Moderna's potential coronavirus vaccine after Dr. Anthony Fauci expressed optimism about the technology behind the biotech's approach.
"I have felt not to get bullish on a vaccine until Fauci said, 'Get bullish,'" said Cramer, suggesting the White House health advisor's positive comments may be that bullish call.
"Fauci's sticking his neck out here. I think it's impressive that he's sticking his neck out," added Cramer, given that Moderna's experimental vaccine platform is still unproven. "I don't want people to get their hopes up, but this would be something."
Fauci, director of the National Institute of Allergy and Infectious Diseases, recently told National Geographic that Moderna's mRNA technology is "really quite impressive." While no vaccines using Moderna's platform have previously been approved, Fauci said it showed "great promise."
Moderna's vaccine candidate uses synthetic messenger RNA to inoculate against the virus. Treatments of this sort help the body immunize against a virus and can potentially be developed and manufactured more quickly than traditional vaccines. The company, like others, leans on artificial intelligence and cloud computing in its vaccine development.
"I was thinking that the way we'd be going was J&J or Pfizer, but if Moderna can do it, you know that we're going higher" in the stock market, Cramer said on "Squawk on the Street."
The race to develop a vaccine for Covid-19 is competitive, but health experts caution that it's an arduous process.
In addition to Moderna, Johnson & Johnson and Pfizer also are working on experimental vaccines. Pfizer, in partnership with German drugmaker BioNTech, has started human testing of its vaccine candidate in the U.S.
Moderna announced on Thursday that the Food and Drug Administration has given it approval to begin phase two trials on the potential vaccine. Moderna said it will commence phase two with 600 participants shortly and is trying to finish plans for a phase three trial as early as this summer. Phase one at Moderna began in the Seattle area in mid-March, the first such trial in the U.S. The Massachusetts-based firm has partnered with the National Institutes of Health.
Fauci and other experts, however, caution that an approved vaccine for wide public use may still be 12 to 18 months away despite the fast-tracking of the trial process.
Nevertheless, Cramer said he found Fauci's comments on Moderna's technology to be a good sign because the infectious disease expert is usually careful with his words. The "Mad Money" host noted that Fauci also spoke optimistically about Gilead Sciences' antiviral drug remdesivir, which has shown early signs of being an effective treatment for Covid-19.

— CNBC's William Feuer contributed to this report.


