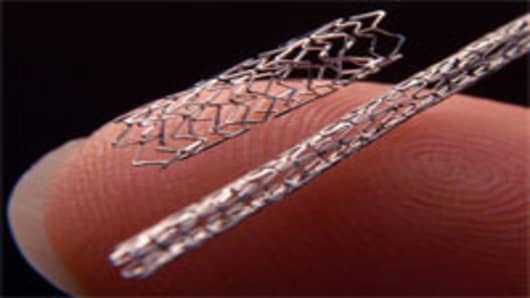
Ahead of the world's biggest gathering of cardiologists in Chicago this weekend, there's a flurry of news about stents--the expensive little wire mesh tubes that prop open clogged arteries.
First, the Food and Drug Administration has posted new proposed guidelines for pre-and post-market testing of the controversial devices. Here's the press releaseand here's the 84-pages worth of draft guidance.
There's a four-month comment period before the FDA might carve the new guidelines in stone. In a research note to clients this morning, Leerink Swann medical device analyst Jason Wittes writes, "The FDA preliminary DES (drug-eluting stent) guidelines came in line with our expectations, with a greater focus on long-term safety and closer assessment of late-stent thrombosis (blood clots that form long after a stent is put in)." Leerink may trade in ABT, BSX and MDT.
Despite the lingering, but less heated debate over how safe and effective they are, stents are a multi-billion-dollar-a-year business. And the U.S. market for stents that are coated with drugs which cut the chances of reclogging is going through a shakeup. Medtronic recently won FDA approval of its first drug-coated stent, destroying the duopoly owned by Boston Scientific and Johnson & Johnson .
And Abbott is waiting for FDA approval of its drug-coated device which BSX will also get to sell under a different brand name. That's part of the weird divestiture terms of BSX's acquisition of Guidant. ABT and MDT will be presenting what Leerink calls "incremental" data on their stents at the Chicago cardiology confab.
But in a separate note to clients today, Leerink's Wittes says he did a "mini survey" of 17 interventional cardiologists--the doctors who put in stents--which indicates Abbott and Medtronic could have a fight on their hands.
The results show that about half of all stent accounts are now locked into long-term contracts, extended one to three years, as BSX and JNJ apparently work to hold onto their corner of the market in the face of the new competition. "We believe this will slow the adoption of MDT's Endeavor (the name of Medtronic's stent) and even ABT's/BSX's Xience/Promus (the two different brand names for their new stent). This is also in stark contrast to years in the past, when stent shares were much more volatile and longer-term contracts less prevalent," Wittes says.
Coincidentally, USA Today has a big article today headlined, "Angioplasty's golden era may be fading" about its own analyses showing that the number of procedures performed in a year has dropped 10 to 15 percent over the past couple of years. That decline started when the first studies appeared about the potential risk of blood clots and then escalated after the report came out last year suggesting old-fashioned heart drugs might work better than stents.
I'll be setting the stage for ACC (American College of Cardiology) Conference in reports on CNBC tomorrow and then reporting live from there all day Monday with reaction to the highly anticipated Vytorin study results and panel discussion about them.
For some interesting photos of stents, please go here.
Questions? Comments? Pharma@cnbc.com