LONDON — As the U.K. rolls out its first coronavirus vaccine to the public, becoming the first country to inoculate people with a fully-tested jab, there are many practical questions about the historic program.
Ninety-year-old Margaret Keenan on Tuesday became the first person in the world to receive the Pfizer-BioNTech vaccine outside of trial conditions. The new shot was approved by the U.K. drug regulator last week.
Here, CNBC answers some of the more popular questions being asked about the vaccine and its rollout:
Why was the UK first to approve the vaccine?
Last Wednesday, the U.K. Medicines and Healthcare products Regulatory Agency (MHRA), became the first regulator in the world to grant emergency authorization to the Pfizer-BioNTech vaccine for use.
When question marks were raised about the speed of the MHRA's authorization, it said it had been carrying out a "rolling review" of the vaccine data as it emerged over the course of clinical trials, and was thus able to make a quick, yet well-informed, decision.
"A dedicated team of MHRA scientists and clinicians carried out a rigorous, scientific and detailed review of all the available data, starting in October 2020," the U.K. government noted in a statement.
"A 'rolling review' can be used to complete the assessment of a promising medicine or vaccine during a public health emergency in the shortest time possible. This is done as the packages of data become available from ongoing studies on a staggered basis," it added.
"The MHRA expert scientists and clinicians reviewed data from the laboratory pre-clinical studies, clinical trials, manufacturing and quality controls, product sampling and testing of the final vaccine and also considered the conditions for its safe supply and distribution."
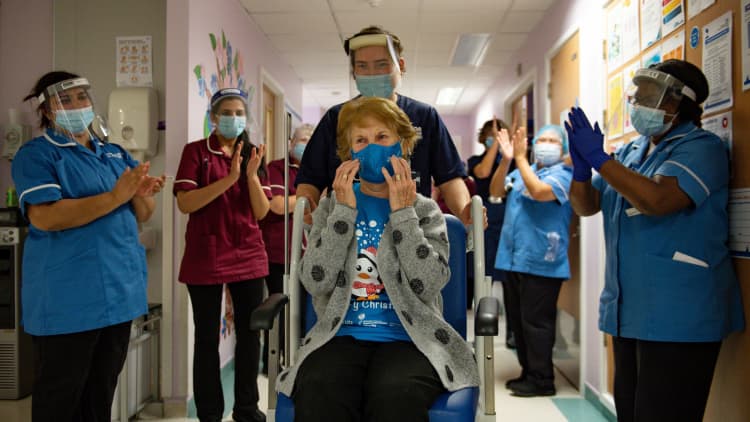
Who made the vaccine?
The new shot in the U.K. is the result of a joint effort between U.S. pharmaceutical giant Pfizer and German biotechnology company BioNTech.
The vaccine that is being supplied to Europe, and all of the U.K.'s 40 million doses (to be delivered in stages with the whole order completed by the end of 2021) come from Pfizer's manufacturing hub in Puurs, Belgium.
How long does the Pfizer vaccine last?
This is the million-dollar question and one that, due to the unprecedented speed of development alongside other coronavirus vaccines, we do not know yet.
Pfizer said in early November, when the companies released their first interim analysis of their Phase 3 clinical trial, that "participants will continue to be monitored for long-term protection and safety for an additional two years after their second dose."
As clinical trials have only taken place this year, it's too early to know the duration of the protection it offers against Covid-19 infection. The coronavirus vaccine, like the flu vaccine, could even become an annual jab, or people could require boosters in future.
Which countries have approved the Pfizer vaccine?
So far only the U.K. has approved the Pfizer-BioNTech vaccine.
The U.S. Food and Drug Administration could make a decision later this week (on Thursday) and the European Medicines Agency has said it will conclude a review into the vaccine by Dec. 29.
On Tuesday, the FDA said the data from Pfizer's trials was consistent with recommendations put forth by the agency for an emergency use authorization.
It also said the vaccine was highly effective and did not raise any specific safety concerns.

Who is getting the vaccine first?
Fifty hospitals in the U.K. are acting as vaccine "hubs" to deliver the shots initially.
Front-line health workers, care home workers and those aged over 80 will receive the vaccine first, before it is given more widely among the U.K. population.
Inoculations will later be given in other community health care centers, like doctors' surgeries.
Which hospitals are getting the vaccine first?
The 50 hospitals in the first wave of vaccinations are:
- Mid and South Essex NHS Foundation Trust
- Milton Keynes University Hospital NHS Foundation Trust
- East Suffolk And North Essex NHS Foundation Trust
- North West Anglia NHS Foundation Trust
- James Paget University Hospitals NHS Foundation Trust
- Norfolk And Norwich University Hospitals NHS Foundation Trust
- Cambridge University Hospital NHS Foundation Trust
- East and North Hertfordshire NHS Trust
- Royal Free London NHS Foundation Trust
- Guy's and St Thomas' NHS Foundation Trust
- Croydon Health Services NHS Trust
- St George's University Hospitals NHS Foundation Trust
- King's College Hospital NHS Foundation Trust
- King's College Hospital NHS Foundation Trust
- Walsall Healthcare NHS Trust
- Chesterfield Royal Hospital NHS Foundation Trust
- Sherwood Forest Hospitals NHS Foundation Trust
- University Hospitals Coventry And Warwickshire NHS Trust
- Royal Stoke Hospital
- Northampton General Hospital NHS Trust
- University Hospitals of Derby and Burton NHS Foundation Trust
- United Lincolnshire Hospitals NHS Trust
- Nottingham University Hospitals NHS Trust
- Shrewsbury And Telford Hospital NHS Trust
- Sheffield Teaching Hospitals NHS Foundation Trust
- North Cumbria Integrated Care NHS Foundation Trust
- Hull University Teaching Hospitals NHS Trust
- The Newcastle Upon Tyne Hospitals NHS Foundation Trust
- Leeds Teaching Hospitals NHS Trust
- South Tees NHS Trust
- Wirral University Teaching Hospital
- Liverpool University Hospitals NHS Foundation Trust
- Countess of Chester Hospital NHS Foundation Trust
- Salford Royal NHS Foundation Trust
- Stockport NHS Foundation Trust
- Blackpool Teaching Hospital
- Lancashire Teaching Hospital Trust
- Frimley Health NHS Foundation Trust – Wexham Park Hospital
- Oxford University Hospitals NHS Foundation Trust
- East Kent Hospitals NHS Foundation Trust – William Harvey Hospital
- Brighton And Sussex University Hospitals NHS Trust – Royal Sussex County Hospital
- Portsmouth University Hospitals Trust
- Royal Surrey NHS Foundation Trust
- Yeovil District Hospital NHS Foundation Trust
- Dorset County Hospital NHS Foundation Trust
- Royal Cornwall Hospitals NHS Trust
- University Hospitals Plymouth NHS Trust
- Great Western Hospitals NHS Foundation Trust
- Gloucestershire Hospitals NHS Foundation Trust
- North Bristol NHS Trust


